Sterilization of water is the process that kills, eliminates or deactivates the all form of microorganisms in the water. It is the critical stage for safe potable water. This method must achieve all most 100% deactivation so that prevents the spread of water-borne diseases. On the other hand disinfection means the process that are removing or reducing harmful microorganisms. There are different methods of sterilization. We can divide them into two methods; chemical methods and physical method.
Chemical methods of sterilization
There are various chemicals are used to the sterilization process. The consumption of these chemical can be reduced by installing a filtration system before this stage. Some of the chemical sterilization methods are described here:-
Ozone sterilization of water
Ozone is a highly effective sterilization chemical for water. It has strong oxidizing properties. It can kills wide range of pathogens microorganisms by oxidizing method. Ozone is more powerful disinfectant than chlorine or chlorine dioxide. Moreover it has effective inactivation activities on giardia or cryptosporidium which is not possible by some others chemicals. This method does not affected by pH. After complete the oxidation process, the excess ozone will breaks into oxygen. So it does not create bad taste or odor’s to the water and leaves no solid residue. On the contrary, it added dissolved oxygen into the water which enhances the water’s taste. The disadvantages of ozone are that it is costly, toxic, unstable and must be produced on mill site. It can create irritation on nose even in low concentrations.
Hydrogen peroxide sterilization
Hydrogen peroxide is another ideal sterilizer like ozone. It is a strong oxidant. It destroys microorganisms without leaving any solid particle to the water. The disadvantage of the hydrogen peroxide is that it is unstable, costly and toxic at high concentrations. The vapor of it can affect the eyes and respiratory system. Some metallic peroxide like Na2O2 has better stability and effectiveness than hydrogen peroxide. Na2O2 produces hydrogen peroxide by the reaction with dissolve carbon dioxide. It also produces sodium carbonate which can able to soften the hard water. You can also apply calcium hydrogen and magnesium hydrogen. Some processes use both hydrogen peroxide and ozone commonly to accelerate the ozone decomposition rate which increases the oxidation rate.
Na2O2 + CO2 + H2O → Na2CO3 + H2O2
Na2CO3 + CaSO4 → Na2SO4 + CaCO3
Chlorination sterilization
Chlorination is the most common and cheapest system for sterilization of water. Chlorine input into the water as chlorine gas, sodium hypochlorite or calcium hypochlorite, chlorine dioxide. When inject it forms several chemicals like hypochlorous acid.
Chlorine sterilization
Chlorine is the most widely used disinfectant in municipal water and wastewater treatment. It can destroy pathogens and control nuisance microorganisms. It can also remove iron, manganese, ammonia nitrogen. Chlorine is a toxic gas so need safety facilities. It acts as a quick oxidizing agent. pH can affects on its activities so needs to control the pH. Chlorine reacts with water and form hypochlorous acid which is further break into nascent oxygen. Both of them are powerful germicide.
Cl2 + H2O → HOCl + HCl ; HOCl → HCl + [O]
Calcium and sodium hypochlorite
Sodium hypochlorite is the one of the most commonly used sterilization chemicals. It is easier and safer to use compared with chlorine gas but more expensive. It is chemically unstable which can convert into sodium chlorate. At higher pH the hypochlorite is corrosive which can attack metal pipe. Calcium hypochlorite can be found as powder, granular, liquid and solid form.
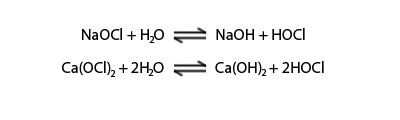
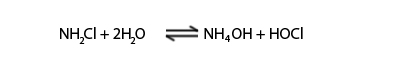
Monochloramine
Monochloramine is another form of chlorination. When chlorine and ammonia included into the water then it forms monochloramine which can react under well controlled conditions. It is poor sterilization chemical compared with chlorine but last long and protect from bacteria for long time.
NH3 + Cl2 → NH4Cl + NH2Cl
Chloramines hydrolysis in water and give HOCl
Chlorine dioxide
Chlorine dioxide is a powerful disinfectant compared with chlorine but more expensive. It must be produced nearby consuming plant site. It is not more used chemical for sterilization of water due to unstable and costly.
Aeration
Aeration can improves water taste and remove iron, Mn, carbon dioxide, H2S, volatile substances and bad odor. There are two type of aeration, natural and artificial. Natural aeration occurs in river, steam. To create artificial aeration several designs like spray is used. During aeration the water absorbs air. So increases the amount of oxygen into the water. This oxygen causes oxidation of the substances which are responsible for forming odor.
Silver ion method
The disinfect effect of copper and silver ions has been known for long time.
The chemical sterilization methods are precipitation method (With alum, lime, soda ash), Potassium permanganate process, Sodium hydrogen sulphate etc.
Physical sterilization methods
Boiling the water
Boiling is one of the oldest and most commonly used water sterilization techniques. If the water is boiled for about 20-30 minutes then it destroy all type of harmful bacteria and algae. They also remove dissolved gases from the water and improve the water taste. This method is only suitable for normal households but not suitable for industrial and large scale purposes.
UV sterilization
Ultra-violet light has a powerful germicidal action on water. It is one of the latest methods for sterilization of water. When ultraviolet radiation is passing through the water then it is absorbed by the cells and damages the DNA of the microorganisms. Hence they are not enable to grow or reproduce. This physical sterilization method can be applied for groundwater and households water but not suitable for large scale. It does not create solid content in water. The water should be free from suspended particles otherwise it may interfere to come in UV rays contract with microorganisms. The system is costly.
Need sterlization / disinfect of 20′ of water pipe at CVC Store HB
Please call John 1-619-838-3969
Ironwood Pldg & Boilers / Santa Ana
job at a cvc store HB
Call asap Sundquistjohn@msn.com